A 63-year-old female with significant PMhx HTN, HLD, IDDM, CAD has been in the hospital for 3 days for uncontrolled hyperglycemia. The patient’s nurse calls you at 8 PM and reports patient appeared acutely altered. You are on your way when the pager goes off, it’s a CODE CVA in that patient’s room. Upon examination, you note that the patient is having trouble with word finding, with slowed speech and diminished sensation on the right. Patient is otherwise oriented x3, able to follow commands, has no other neurologic deficits. Patient was apparently interactive and resting comfortably at 7 PM. Yes, her fingerstick is good at 130 mg/dL. At this time, your concern is obviously high for a CVA and you proceed with the CVA work up to identify whether patient did, in fact, have a CVA.
So how do you respond to this ‘code CVA’?
In this post, we will focus on the diagnostic evaluation for ischemic CVA’s and on the immediate management in a future part II of this post.
What are the types of strokes?
Prior to diving in, here is a brief overview of cerebrovascular accidents (CVA). The event typically presents as an abrupt onset of neurologic impairment due to a vascular insult. According to the American Stroke Association, there are three main types of presentations that guide work up and management:

Briefly, in ischemic strokes, severe focal hypoperfusion leads to free radical formation and microvascular injury. Once the initial injury to the area occurs, it leads to inflammatory changes which can worsen the initial area of damage, leading to more permanent damage, known as infarction. Infarction, the hallmark of ischemic strokes, causes irreversible neuronal injury at the core of the area affected. Acknowledge that even TIAs, once considered reversible cerebral ischemia given transient neurologic symptoms, can still cause permanent injury, i.e. infarction. Surrounding this area of ischemia/infarction is the penumbra – which can be salvaged if the blood flow returns. While anatomically intact, the penumbra requires higher energy consumption to depolarize due to this developing ischemic cascade. The penumbra is the area of the brain that we aim to salvage with prompt restoration of blood flow and permissive hypertension (see future post).

It is important to understand the pathophysiology because the next step is to figure out how the event occurred, thus allowing us to classify it, and then allowing us guidance on appropriate management.
Now back to responding to that ‘Code CVA’.
How do you evaluate a patient in a ‘Code CVA’?
You are the first to the room. You remain calm and you follow your usual checklist. ABCs. IV. Monitor. And so on.
- ABCs– ensure they are hemodynamically stable and able to protect their airway. Do not forget your ABCs.
- Ensure adequate IV access. Make sure the patient is connected to a telemetry monitor. Then proceed.
- Obtain a finger stick glucose to rule out hypoglycemia. Ask about recent medication administrations.
- Physical exam – in addition to your usual physical exam, complete a thorough neurologic examination, specifically one catered to stroke evaluation.
- Pro tip #1: check out Dr. Di’s neurologic examination template
- Pro tip #2: use a risk stratification tool, as described below, to ensure a complete stroke evaluation.
- Pro tip #3: be wary of prior neurologic deficits (weakness, hemi- or quadriplegia, etc) or even a language barrier.
- EKG – obtain an electrocardiogram (see below).
- If exam raises concern for CVA, get a CT head without contrast (NCCT). The goal is to RULE OUT hemorrhagic CVA and other mimics (such as tumors), not to diagnose an ischemic CVA or TIA. So, if they have focal deficits and your concern in high based on exam and risk stratification tools, obtain a CT head immediately.
- While an NCCT takes approximately 6 to 8 hours to identify an ischemic stroke and will likely not show ischemic changes, they still can be picked up on NCCT (see below).
What are some scoring systems for risk prediction and severity?
There are several stroke diagnostic scales such as Fast Arm Speech (FAST), Recognition of Stroke in the Emergency Room (ROSIER), and Rapid Arterial oCclusion Evaluation (RACE). The ROSIER and FAST scales have similar sensitivity, greater than 80%, but poor specificity. The RACE scale is based on the National Institute of Health Stroke Scale (NIHSS).
FAST assesses Facial weakness, Arm weakness, Speech Impairment and Time. The BE-FAST considers Balance and Eyes – visual impairments, but it has not yet been validated.
ROSIER takes into account any loss of consciousness or syncope as well as seizure activity. On examination, it accounts for facial weakness, arm weakness, leg weakness, speech disturbance and visual field deficits.
RACE assesses facial palsy, arm motor weakness, leg motor weakness, head and gaze deviation, aphasia and agnosia. A RACE score of ≥5 had sensitivity of 85% for identifying large artery occlusions (LAOs); it has the highest predictive value for LAOs.
Finally and most importantly, the NIHSS is a 15-item stroke impairment scale validated in clinical trials and patient care. There is a high degree of reliability in using the NIHSS due to the standardization that is available through video training for both physicians and nurses. The NIHSS is scored from 0-42. While it is standardized, keep in mind:
- a score of 1 for sensory impairment is very different from a score of 1 for aphasia. One is a minor impairment with no disability whereas aphasia will certainly have long term consequences and intervention is warranted
- it does not account for all stroke impairments, like a posterior circulation stroke involving the brainstem
- the NIH Stroke Scale website provides guidance when needing to account for prior neurologic deficits, language barriers, being intubated, etc.
If you decide based on risk stratification and physical exam that the patient’s change is concerning for a CVA, proceed with the ‘CODE CVA’, a formal organized protocol where
- someone from your team or you will follow the patient until completion of the protocol (yes, go with them to the CT scan),
- and at our institution, the patient will have a video evaluation with a tele-neurologist, who will guide additional testing and management (hint hint: tPA or no tPA).
What are we looking for in a diagnostic workup?
We are trying to 1) obtain radiographic evidence of a stroke and 2) determine the underlying cause. These causes can be classified by utilization of the TOAST (est 1993) or ASCO (est 2009) classification. TOAST attempted to characterize strokes by the most likely cause(s) of ischemic stroke: small vessel disease, large vessel disease, cardioembolic stroke, cryptogenic and strokes of other etiologies. ASCO was developed to introduce ‘stroke phenotyping’, i.e. the etiology + presence of underlying disease + severity. It includes (A)therosclerosis, (S)mall vessel disease, (C)ardiac disease and (O)ther causes along with a grading of 1 (definitely a potential cause of the stroke), 2 (causality uncertain), 3 (unlikely a direct cause of the stroke but disease is present). When the disease is completely absent, the grade is 0.
What testing do you obtain?
Step 0 (ie your first step) is to rule out a bleed, not confirm ischemic CVA. It also helps exclude stroke mimics such as from a tumor. The non-contrast CT (NCCT) has limited sensitivity in the acute setting for picking up ischemic changes. It does, however, help you exclude other intracranial pathologies that may mimic a stroke. Interestingly, in some institutions, MRI is used as the initial imaging modality given greater Sn/Sp and CT imaging is skipped altogether; for obvious reasons this does not apply at most institutions.
Step 1: Confirm ischemic CVA
- NCCT: this may be beneficial in identifying large infarctions originating from the MCA. Specifically, for strokes caused by MCA occlusions, a hyperdensity of an artery (hyperdense vessel sign), loss of gray-white matter differentiation, cortical sulcal effacement or changes of the Sylvian fissure are representative of early infarcts. The Alberta Stroke Program Early CT Score (ASPECTS ) evaluates 10 different areas supplied by the MCA for these changes. A similar Posterior Circulation ASPECT Score (PC-ASPECTS) has been developed for strokes involving the posterior circulation.
- MRI brain without contrast: this is the gold standard imaging modality. Protocols that combine DWI and FLAIR are the currently recommended modality to identify an ischemic stroke within minutes of onset. It is the most sensitive, specific and accurate in detecting ischemic core. Keep in mind:
- there are no studies to show MRI is superior to CT for identifying patients who require intervention with alteplase
- MRI is also limited at most facilities and may not be readily available, delaying diagnosis and treatment. A lot of patients also cannot tolerate MRI due to increased scan time.
3. CT angiography +/- CT perfusion: This will help identify an occluded vessel, AVMs, dissection and other vascular pathologies. It can detect occlusions in the carotids as well. Also, a CT angiogram can be completed much faster and is more readily available at most institutions than MRI. CT perfusion (CTP) imaging follow the contrast from CTAs into brain parenchyma and analyze tissue perfusion through analytical software. It gives us an idea of the size of the ischemic core and of tissue that is critically hypoperfused. At our institution, MRI is available in 24 hours but CT perfusion is not.
- Per Ezzeddine et al., CT angiography and CT perfusion are more accurate in identifying acute ischemic stroke. They more readily identify the location of vascular compromise and infarction.
- A more recent study by Campbell et al., made the same determination regarding CT perfusion. As compared to MRI, which requires patient to lay still for a prolonged period as well as cause claustrophobia, CT perfusion and CT angiogram took 12 minutes on average, to complete and read. Both studies are beneficial for rapid diagnosis of suspected stroke.
- If available, consider early CT angiography for faster identification of vascular compromise requiring intervention.
Step 2: Rule out small vessel disease
- Lacunar strokes will be visualized on either of above imaging modalities but are best identified on an MRI without contrast.
Step 3: Rule out large vessel disease
There are 2-separate considerations to this: intracranial and extracranial.
Intracranial is discussed above in step 1. The only other only imaging modality to add is Transcranial doppler (TCD) which uses ultrasound to insonate large intracranial vessels for stenosis and collaterals on a real-time basis. The obvious drawback / limitation would be examiner-dependence, lack of sufficient experienced ultrasonographers and poor sensitivities in certain vessels.
Extracranial: evaluate for stenosis or occlusion in the neck vasculature via CTA/MRA neck [NA1] or carotid ultrasonography.
- Carotid ultrasound is a useful and cheap modality to screen patients with suspected stroke, to identify any large vessel disease in the anterior neck vasculature (ex/ internal carotids). However, it does not allow for sufficient posterior vessel imaging (vertebrobasilar, etc) and in an acute setting when time is of the essence, CTA and MRA are more standardized. In the setting of vertigo, vision finding difficulty or other evidence of posterior circulation compromise, CT/MR neck [NA2] angiography is the preferred imaging modality and therefore, carotid US is not needed.
- For patients without previous work up, obtain a lipid panel and hemoglobin A1c as both are good indicators for likelihood of the presence of atherosclerotic disease.
Step 4: Rule out cardioembolic source
- Arrhythmia such as atrial fibrillation causes a large percentage of ischemic strokes. Therefore, electrocardiogram at the time of event as well as further telemetry monitoring for at least 24 hours is recommended. If negative and still suspecting arrhythmia, consider a Holter monitor. In elderly patients, with high suspicion for arrhythmia, consider loop recorder.
- Ideally, a transthoracic echo (TTE) can identify cardioembolic sources such as a clot in the left atrium or an LV thrombus. If really concerned, it may be followed up with a transesophageal echo (TEE) after discussion with cardiology colleagues. If younger than 60, consider adding on a bubble study to either as incidence of patent foramen ovale is high (1/4 patients) and can be considered a precipitating factor.
Step 5: Rule out other causes
- In younger patients without risk factors, consider a hypercoagulability workup. No, not every patient should be worked up for hypercoagulability. However, every patient should have a CBC and coagulation studies to rule out pro-thrombotic states and etiologies such as polycythemia, thrombocytosis, leukemia.
- Your vascular imaging should rule out carotid or vertebral artery dissection, carotid artery stenosis and malformation, for whichcerebral angiography (catheter based vessel mapping) is gold standard for imaging carotid artery stenosis, vasculitis, malformations and aneurysms.
- Consider vitamin B12 levels, TSH and RPR. While these lab abnormalities can mimic an acute stroke, first identify and rule out an acute event. These tests have very low yield.
Step 6: If the work up turns out wholly negative, consider cryptogenic stroke
- Cryptogenic stroke is defined as a stroke without a clearly identifiable source and can be differentiated as embolic versus non-embolic.
- Embolic Stroke of Undetermined Etiology (ESUS) is a type of cryptogenic stroke and refers to ischemic strokes with embolic findings. ESUS was coined in 2014 to refer to non-lacunar infarct with the absence of an identifiable source. Patients with ESUS typically lack risk factors but their risk of recurrence remains high, approximately 5% per year, per a recent JACC review.
- Examples of cryptogenic stroke include patent foramen ovale, ventricular septal defect, atherosclerotic disease of the aorta and valvular heart disease.
What about TIAs?
If the imaging work up was negative but your risk stratification and exam showed high probability of CVA, it was likely a resolved ischemic event. Management aside (see future post), diagnostic evaluation for TIAs involves additional pretty much the same steps, such as lab work, echocardiography and carotid ultrasound. TIAs can be risk stratified using the ABCD2 score which determines risk of stroke after TIA. An MRI will show chronic white matter changes, indicative of higher risk of future ischemic events.
In Summary:
- A CVA is an abrupt onset of neurologic impairment due to vascular insult. Ischemic CVA is the most common, etiologies are determined by the TOAST classification – small vessel disease, large vessel disease, cardioembolic causes, other causes and cryptogenic strokes.
- A thorough evaluation, including ABCs and physical examination, is required prior to proceeding with a CODE CVA. Obtain an NIHSS score and use one of the risk stratification scores while evaluating the patient initially, if high suspicion. Follow alongside the patient through the entire protocol.
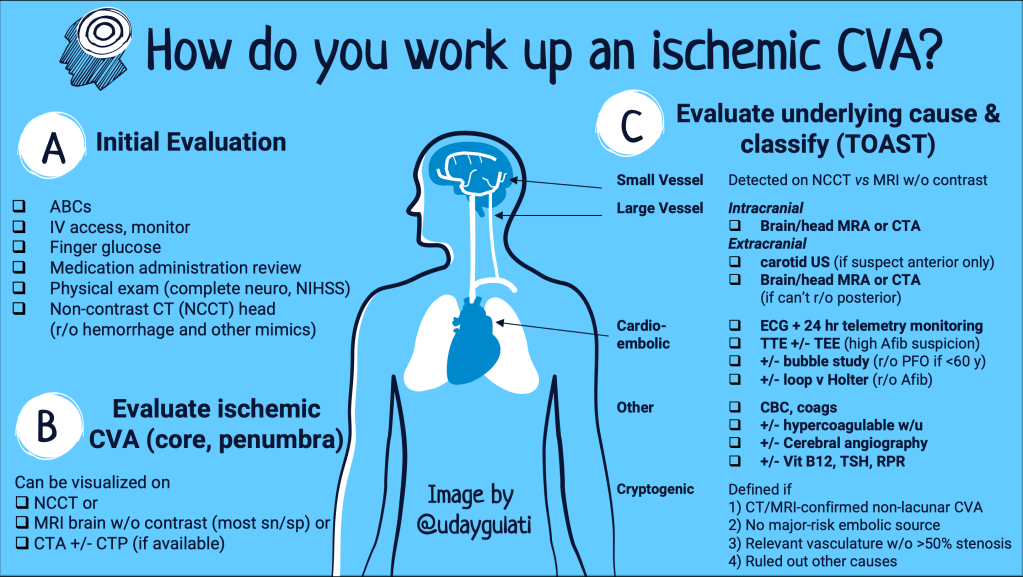
- Non-contrast CT head is used to RULE OUT hemorrhage, not diagnose ischemic stroke.
- At our institution, further evaluation is completed with MRI without contrast given high Sn/Sp and ready availability. For large vessel imaging of the brain head, we don’t have CT perfusion imaging so use the CTA or MRA brain/head. CT angiography is faster to complete and more readily available at most hospitals and can be obtained alongside the NCCT during initial imaging.
- For large extracranial imaging, carotid ultrasound can be used to identify problems with anterior circulation but if there is concern for posterior circulation stroke, a CTA or MRA is better suited, at which point the carotid US is not needed.
- Lab work including CBC, coagulation studies, HbA1c, and lipid panel are indicated for every patient.
Post reviewed and edited by @udaygulati
Sources:
Birenbaum D, Bancroft LW, Felsberg GJ. Imaging in acute stroke. West J Emerg Med. 2011;12(1):67-76.
Brinjikji W, Rabinstein A, Cloft HJ, Lanzino G, Kallmes DF. Recently Published Stroke Trials: What the Radiologist Needs to Know. Radiology. 2015;276(1):8-11. doi:10.1148/radiol.2015150422
Campbell BC, Weir L, Desmond PM, et al. CT perfusion improves diagnostic accuracy and confidence in acute ischaemic stroke. J Neurol Neurosurg Psychiatry. 2013;84(6):613-618. doi:10.1136/jnnp-2012-303752
Ezzeddine MA, Lev MH, McDonald CT, et al. CT angiography with whole brain perfused blood volume imaging: added clinical value in the assessment of acute stroke. Stroke. 2002;33(4):959-966. doi:10.1161/hs0402.105388
Hui C, Tadi P, Patti L. Ischemic Stroke. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2020.
Lakhan SE, Kirchgessner A, Hofer M. Inflammatory mechanisms in ischemic stroke: therapeutic approaches. J Transl Med. 2009;7:97. Published 2009 Nov 17. doi:10.1186/1479-5876-7-97
Nouh A, Hussain M, Mehta T, Yaghi S. Embolic Strokes of Unknown Source and Cryptogenic Stroke: Implications in Clinical Practice. Front Neurol. 2016;7:37. Published 2016 Mar 21. doi:10.3389/fneur.2016.00037
Todo K, Sakai N, Kono T, et al. Alberta Stroke Program Early CT Score-Time Score Predicts Outcome after Endovascular Therapy in Patients with Acute Ischemic Stroke: A Retrospective Single-Center Study. J Stroke Cerebrovasc Dis. 2018;27(4):1041-1046. doi:10.1016/j.jstrokecerebrovasdis.2017.11.007
Wey HY, Desai VR, Duong TQ. A review of current imaging methods used in stroke research. Neurol Res.2013,35(10): 1092-1102. doi: 10.1179/17431132813Y.0000000250.




Thanks for including information about cryptogenic strokes. My brother has valvular heart disease (https://www.premiermedicalhv.com/divisions/services/valvular-heart-disease/) and he had a cryptognenic stroke a few years back.
LikeLike