A 56-year-old woman with a history of hypertension and COPD experiences a sharp chest pain and acute dyspnea. An emergency medical team was activated, was found to be in Ventricular Fibrillation and the VF was successfully defibrillated with an automatic external defibrillator. The patient was taken to a cardiac care unit by the emergency medical team where she received a full workup to rule out coronary artery disease, cardiomyopathies and channelopathies. The team determines she has severe ischemic cardiomyopathy. Should the patient receive an implantable cardioverter-defibrillators (ICD)?
Let us review.
What is an AICD?
An AICD is an automated implantable cardioverter-defibrillator device (or simply ICD) used typically in heart failure (HF) patients for cardiac rhythm management. They even have use for patients with unexplained syncopal episodes that meet certain criteria. These devices are designed to recognize and terminate life-threatening ventricular arrhythmias. The term automated simply means that the process of recognition and subsequent termination is all automatic within the device itself inside patient’s chest.
The manual process that we are all aware of occurs whenever a patient has circulatory collapse and/or cardiac arrest, and the proverbial defibrillator “paddles” are put on the patient’s chest while the doctor charges the machine, and then pushes a button to deliver an electric shock to the patient’s heart in an effort to restore NSR.
There are a few different types of AICDs:
- single chamber with one lead,
- dual chamber with two leads,
- and biventricular ICDs with three leads used for cardiac resynchronization therapy (CRT).
The purpose here is to gain a general sense of AICD form and function, their importance, and indications for placement mainly for primary prevention of sudden cardiac death (SCD) in HF patients. The complexities of the AICD types/functions including biventricular ICDs for CRT won’t be discussed here.
Why are they important?
AICDs can provide both primary and secondary prevention and quite a few RCTs have demonstrated their mortality benefit in terms of improved overall survival compared to antiarrhythmic medications alone in patients at risk for SCD. SCD here means death due to ventricular tachycardia (VT) or ventricular fibrillation (VF). Remember, VT can degenerate or progress to VF and patients with reduced LVEFs and/or prior MI are at risk for developing SCD.
CAD patients with associated cardiomyopathy (ICM) who meet certain criteria are at risk for SCD, and implantation of an AICD can potentially prevent SCD from occurring in the first place. This also has application in patients with nonischemic cardiomyopathy (NICM) as there is positive data supporting their use in this patient population too.
Wouldn’t you want an implantable device that can deliver synchronized cardioversion in VT or deliver defibrillator doses of electric current to your heart should you be unlucky enough to go into one of these life-threatening arrhythmias? It’s about risk reduction in terms of mortality and the data is pretty clear. Let’s dive in.
What is the supporting evidence?
The SCD-HeFT trial (published in 2005, NEJM) showed a survival benefit in HF patients with an AICD in place compared to patients without an AICD on just amiodarone or placebo. In fact, this trial included both ICM and NICM patients. The primary outcome was all-cause mortality.

Image Credit: SCD-HeFT trial published NEJM, 2005
Another study called the MADIT-II trial (published 2002, NEJM) looked at post-MI patients with reduced LVEFs and randomized them to either prophylactic AICD placement vs. conventional medical therapy and then looked at the difference in all-cause mortality (primary outcome). The AICD group did better here as well (14.2% vs. 19.8%, p=0.016).
As a result of these trials and others, there is a Class I recommendation from the 2017 AHA/ACC/HRS guideline for AICD placement in HF patients with reduced LVEFs meeting certain criteria for primary prevention of SCD.
So what are the indications for AICD placement?
The focus here is primary prevention of SCD in CAD/HF patients. Selection of patients for AICD placement has to be individualized but the following criteria should generally be met when considering placement:
- LV-EF 35% or less
- GDMT: Be on guideline-directed medical therapy (GDMT) – essentially all of the appropriate HF meds we use like ACE-I or ARBs, BB, Aldactone, and/or BiDil if necessary
- NYHA: functional status of NYHA class II or III HF
- Timing: Be at least 40 days post-MI or at least 90 days post-revascularization procedure (stents/CABG)
- Survival: Have an expected survival > 1 year
If you enjoy mnemonics,
Low EF Gonna Need The Shock
for “LV EF, GDMT, NYHA, Timing and Survival”
If your CAD patient meets the above criteria and he or she is agreeable to AICD placement then it has to considered because you’re effectively reducing the patient’s mortality risk from SCD, and therefore, increasing the patient’s chances at survival.
If you enjoy more detail, the American Heart Association (AHA) summarize their recommendations in these patients with the following indications:
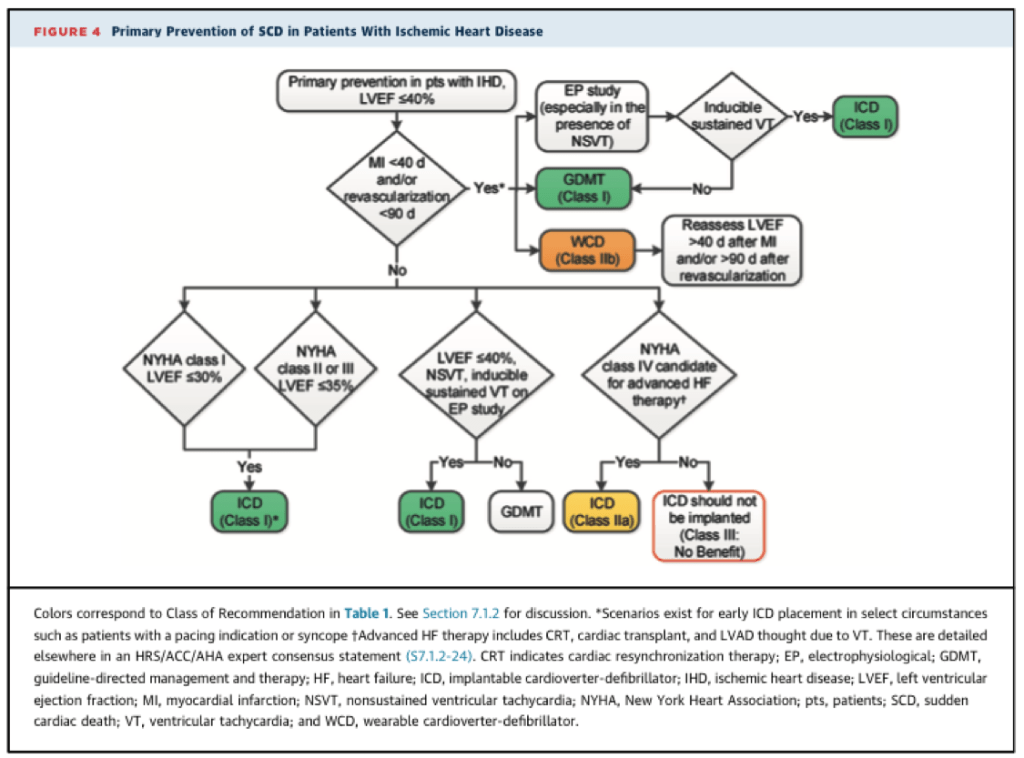
It should be noted that the 40-day post-MI wait has to do with the fact that trials looking at AICD placement earlier than 40 days did not show any benefit. The 90-day wait allows time for recovery of LV function if any is going to take place while the patient is on appropriate GDMT, which is important to note because ACE-I and BBs in particular can actually improve LVEF if the patient is compliant. If on reassessment the LVEF is still reduced 90 days out, then AICD placement is warranted in order to best minimize the risk of SCD. This is often the time period where we see patients with a wearable cardioverter-defibrillator, which we typically know as the Life Vest.
In summary…
AICDs are life saving devices with a proven mortality benefit for patients with HF with reduced ejection fraction (HFrEF).
The Journal of the American College of Cardiology (JACC) provides this neat checklist to review when determining need for AICDs:

AICDs are a broad topic with even more indications but the points above give us some knowledge about their specific utility and what to look for in our HF patients with reduced EF. In that way, we can best do our part to decrease morbidity and mortality in this particular patient population.
References
- Al-Khatib SM, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society
- Bardy GH, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. The New England Journal of Medicine. 2005
- Moss AJ, et al. Prophylactic Implantation of a Defibrillator in Patients with Myocardial Infarction and Reduced Ejection Fraction. The New England Journal of Medicine. 2002.
- http://www.onlinejacc.org/guidelines/VASCD
Post reviewed and edited by @larrybuck2




